ARA-290 (15mg)
$95.00
ARA-290 is a non-erythropoietic peptide derived from erythropoietin, designed to retain the tissue-protective and anti-inflammatory properties of erythropoietin without stimulating erythropoiesis. It has shown promise in various therapeutic applications due to its ability to modulate immune responses and protect tissues.
ARA-290 Product Description
ARA-290 is a non-erythropoietic peptide derived from erythropoietin, designed to retain the tissue-protective and anti-inflammatory properties of erythropoietin without its hematopoietic effects. This makes ARA-290 a promising candidate for research in various medical conditions where inflammation and tissue damage are prevalent.
One of the key areas of interest is its potential in treating neuropathic pain and metabolic disorders. In patients with type 2 diabetes, ARA-290 has shown promise in improving metabolic control and alleviating neuropathic symptoms, suggesting a role in both metabolic and nerve health.
Peptide Information
| Property | Value |
|---|---|
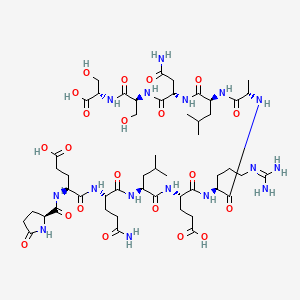
| Peptide Sequence | pGlu-Glu-Gln-Leu-Glu-Arg-Ala-Leu-Asn-Ser-Ser |
| Molecular Formula | C51H84N16O21 |
| Molecular Weight | 1257.3 g/mol |
| CAS Number | 1208243-50-8 |
| PubChem CID | 91810664 |
| Synonyms | Cibinetide, 1208243-50-8, ARA290, PHBSP, ARA 290 |
ARA-290 Structure
Source PubChem
ARA-290 Research
ARA-290 is a novel peptide derived from erythropoietin (EPO) that has emerged as a promising therapeutic candidate across multiple disease areas. Unlike its parent molecule EPO, ARA-290 selectively activates tissue repair mechanisms without stimulating erythropoiesis, offering a potentially safer therapeutic profile. This synthetic peptide mimics EPO’s tissue-protective properties by specifically binding to the innate repair receptor (IRR), a distinct receptor complex that mediates cytoprotective and anti-inflammatory effects.
Initial preclinical and clinical studies have demonstrated ARA-290’s broad therapeutic potential, particularly in conditions characterized by inflammation, tissue damage, and neuropathy. Its unique mechanism of action involves modulating immune responses, promoting tissue repair, and protecting neural tissue, making it particularly relevant for diseases ranging from neurodegenerative conditions to metabolic disorders.
ARA-290 and Alzheimer’s Disease
ARA-290 has been investigated for its potential in Alzheimer’s disease (AD) therapy. It was found to decelerate the progression of AD-like pathology in early-onset models by modulating monocyte functions, specifically increasing the ratio of patrolling monocytes that help clear amyloid-β from the brain.1
Type 2 Diabetes and Neuropathy
ARA-290 has been evaluated in patients with type 2 diabetes, where it improved metabolic control and neuropathic symptoms. It was shown to enhance hemoglobin A1c levels and lipid profiles, as well as increase corneal nerve fiber density, indicating its potential in managing diabetes-related complications.2
Pain Management
In patients with sarcoidosis-associated small fiber neuropathy, ARA-290 has been effective in reducing neuropathic pain and improving quality of life. It increased corneal nerve fiber density and improved sensory pain thresholds, suggesting its role as a disease-modifying agent.3
ARA-290 also exhibits analgesic properties by targeting the TRPV1 channel, which is involved in pain sensation. This mechanism highlights its potential as a novel pain treatment option, integrating immune modulation with nociception.4
Autoimmune Neuropathies
In experimental autoimmune neuritis, ARA-290 has shown efficacy in suppressing inflammation and promoting nerve regeneration without inducing hematopoiesis, making it a candidate for treating autoimmune neuropathies.5
Tissue Repair
ARA-290 has been studied for its tissue-protective effects in critical limb ischemia, where it improved functional and histological outcomes in ischemic conditions, supporting its development as a therapeutic adjunct for ischemic injuries.
ARA-290 (12037)

ARA 290 (260256)

ARA-290 (251426)

Be the first to review “ARA-290 (15mg)” Cancel reply
Related products
Vials
Vials
Vials
Vials



Reviews
There are no reviews yet.